Autoimmune
Stem Cell Treatment
Stem Cell Therapy for Lyme Disease: What the Science Actually Says
Can mesenchymal stem cells help with chronic Lyme disease? A deep dive into the biology of post-treatment Lyme, the immune dysfunction that drives it, and why we offer this treatment at our clinic.
"Antibiotics cure Lyme disease."
That statement is mostly true. And it's precisely where the problem begins.
Because for an estimated two million Americans, the antibiotics worked. The Borrelia burgdorferi bacteria were eliminated. The blood tests came back clean.
And yet the fatigue stayed. The joint pain stayed. The brain fog stayed.
This is post-treatment Lyme disease syndrome (PTLDS)—and if you or someone you love is living with it, you already know: being told you're "cured" when you still can't think straight is its own particular kind of frustrating.
We see these patients at our clinic every week. People who've done everything right—caught it early, finished their antibiotics, followed up with their doctors—and are still struggling months or years later.
So here's the question: if the bacteria are gone, what's still causing the symptoms?
And more importantly—can stem cells do anything about it?
Let's walk through the data.
Roadmap
- What actually causes PTLDS?
- How Borrelia breaks your immune system on the way out
- What mesenchymal stem cells (MSCs) do—mechanistically
- What the clinical evidence shows (and where it's thin)
- What treatment at our clinic looks like
- Safety, cost, and honest limitations
What Actually Causes PTLDS?
This is the key question. And for a long time, medicine didn't have a great answer.
The conventional framing was simple: antibiotics kill the bacteria, symptoms resolve, done. If symptoms persisted, the assumption was either incomplete treatment or psychosomatic causes.
That framing has aged poorly.
A 2020 study in the Journal of Clinical Investigation identified that PTLDS involves distinct pathogenic mechanisms driven by maladaptive host immune responses. Translation: your immune system keeps firing even after the target is gone.
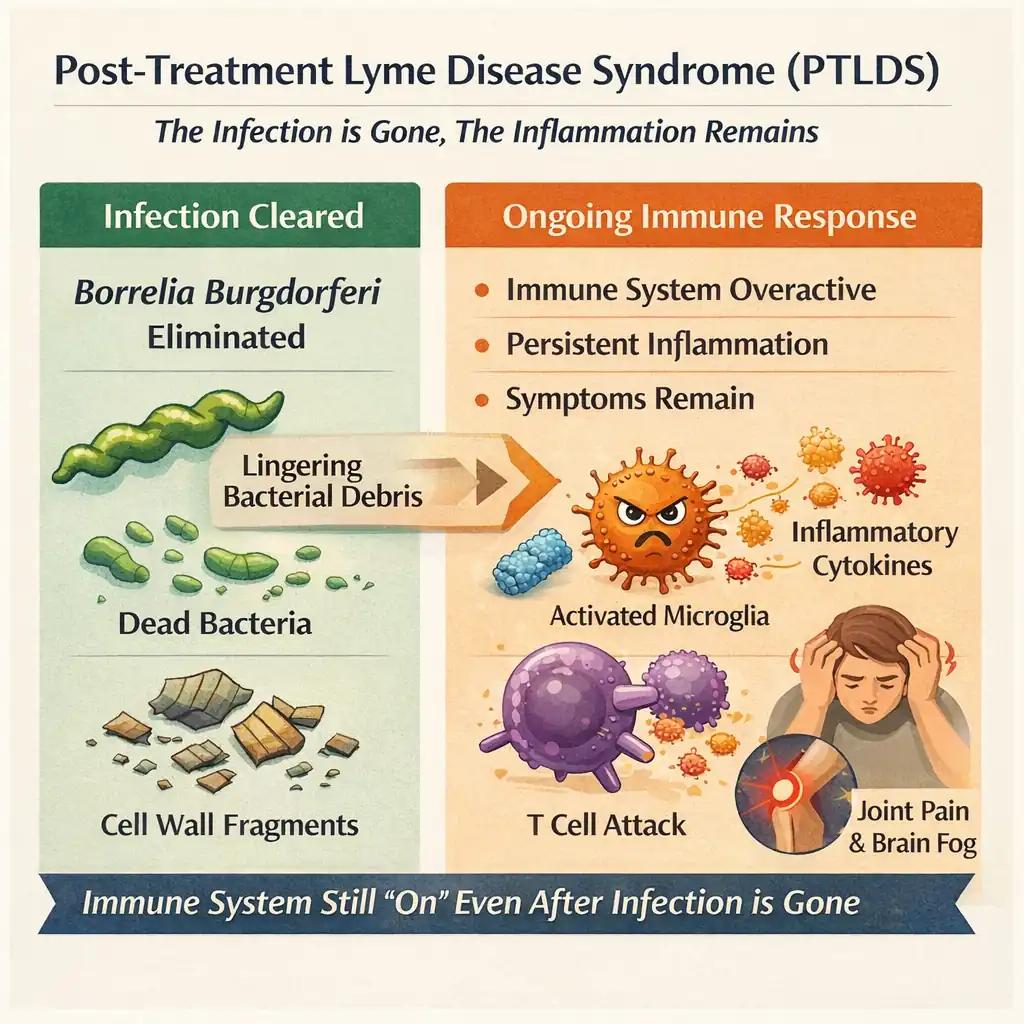
Then in 2025, researchers at Northwestern's Feinberg School of Medicine found something that shifted the conversation further. Fragments of Borrelia's cell wall—called peptidoglycan—persist in the body for weeks to months after antibiotic treatment.
Let that land for a second.
The living bacteria are dead. But pieces of their architecture remain. And those remnants keep triggering your immune system like a car alarm that won't shut off.
This is the paradox at the heart of chronic Lyme:
The infection is gone. The disease remains.
And this is exactly why we started offering stem cell therapy for PTLDS patients. Because once you understand the problem at this level, the standard playbook—more antibiotics, symptom management, wait and hope—starts to look insufficient.
How Borrelia Breaks Your Immune System
To understand why the aftermath is so bad, you need to understand how Borrelia burgdorferi fights on the way in.
This is not a dumb bacterium. Research published in Frontiers in Immunology (2017) catalogued its evasion toolkit, and it's genuinely impressive:
- Antigenic variation (VlsE): Borrelia reshuffles its surface proteins constantly. It's like changing disguises faster than your antibodies can issue a wanted poster.
- Complement hijacking (CRASPs): It deploys specialized surface proteins that essentially recruit your own complement system—your immune network's first line of chemical defense—and turn it off.
- B cell suppression: It interferes with the class-switching process that normally sharpens your antibody response over time.
The result? An immune system that's simultaneously overstimulated and misdirected.
It's attacking everything. It's hitting nothing precisely. And the collateral damage—to your joints, your nerves, your brain—accumulates.
Even when the bacteria are cleared, the immune system doesn't just snap back to normal. It stays dysregulated. The inflammation persists. The tissue damage compounds.
This is what PTLDS actually is. Not a lingering infection. A broken thermostat.
Enter Mesenchymal Stem Cells
So here's the logic: if the core problem in PTLDS isn't bacteria but immune dysfunction, then maybe the intervention shouldn't be another antibiotic.
Maybe it should be something that retrains the immune system itself.
That's the premise behind mesenchymal stem cell (MSC) therapy—and it's why we've built our Lyme disease treatment protocol around it.
Let's be clear about what MSCs are not. They're not going to transform into new neurons or rebuild your knee cartilage in meaningful numbers. That's not how they work.
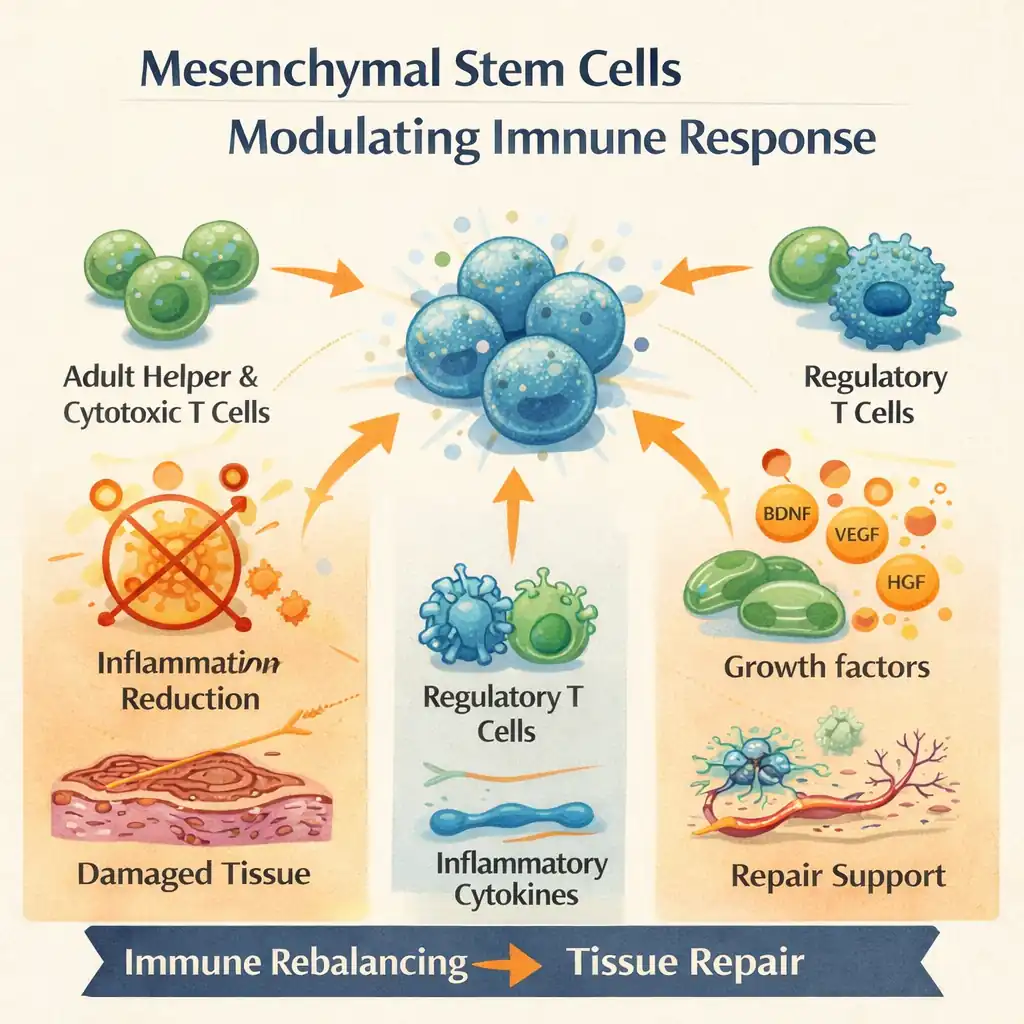
What they actually do is communicate.
MSCs are essentially cellular diplomats. They interact with your immune cells—T cells, B cells, macrophages, natural killer cells—and modulate their behavior. They don't fight the disease directly. They change the environment in which your immune system operates.
Here's the mechanistic breakdown:
Immune rebalancing. MSCs expand the population of regulatory T cells (the peacekeepers) while suppressing overactivated helper and cytotoxic T cells (the ones doing collateral damage). Research in Stem Cell Reviews and Reports (2023) confirmed that MSCs inhibit pro-inflammatory pathways while stimulating anti-inflammatory ones.
- This directly addresses the core immune dysregulation in PTLDS.
Inflammation control. MSCs secrete anti-inflammatory molecules—IL-10, TGF-β, prostaglandin E2—that reprogram macrophages from a destructive pro-inflammatory state to a reparative one. A 2022 study in the International Journal of Molecular Sciences mapped this macrophage reprogramming in detail.
- Think of it as switching your immune cells from "attack mode" to "repair mode."
Tissue repair. MSCs release growth factors—BDNF for neurons, VEGF for blood vessels, HGF for tissue regeneration—that create an environment where damaged tissue can actually heal.
- They don't rebuild your tissues. They create the conditions under which your tissues can rebuild themselves.
That distinction matters. And it's one we make sure every patient at our clinic understands before treatment.
The Evidence: What's Strong, What's Thin
I'll be transparent here, because this is where things get nuanced—and where we think clinics owe their patients honesty rather than hype.
There are no large-scale, randomized, double-blind trials of MSC therapy specifically for Lyme disease.
That's a meaningful gap. It doesn't invalidate the approach—but it means we're working from adjacent evidence. And you should know that going in.
Here's what does exist:
- Rheumatoid arthritis: A 2023 systematic review in PLOS One confirmed both safety and efficacy of MSC therapy in reducing inflammatory markers across multiple controlled trials. RA shares core inflammatory mechanisms with chronic Lyme.
- Multiple sclerosis: A Phase II randomized, placebo-controlled trial in Stem Cell Research & Therapy (2024) showed promising results for MSC-derived therapy in progressive MS—a condition whose immune-mediated neurological damage mirrors neuroborreliosis.
- Inflammatory arthritis broadly: A 2024 meta-analysis in Seminars in Arthritis and Rheumatism analyzed 36 randomized trials, 2,076 participants. Findings supported the efficacy and safety of MSC therapy for inflammatory joint conditions.
- COVID-19 immune dysregulation: A 2024 meta-analysis in the Journal of Translational Medicine found MSC treatment significantly reduced mortality and inflammation—without increasing adverse events.
- Chronic Lyme specifically: A study from Rowan University evaluated MSCs as an adjuvant therapeutic specifically for chronic Lyme disease—one of the first direct investigations connecting MSC science to the PTLDS population.
And a milestone worth noting: on December 18, 2024, the FDA approved the first mesenchymal stem cell therapy for commercial use. As Signal Transduction and Targeted Therapy (2025) reported, 39 clinical studies have now investigated the immunomodulatory applications of MSCs across infections, autoimmune diseases, and inflammatory disorders.
The picture is incomplete. But it's not blank. And what's there is strong enough that we feel confident offering this to patients—while being upfront about exactly where the evidence stands.
The Stem Cell Decline Problem
One more piece of context that's worth understanding—and one that comes up constantly in our consultations.
By age 35, you've lost roughly 5–10% of your original stem cell count. By 50, that's 30–50%. By 70, you're down to 10–30% of what you started with.
This matters for Lyme specifically.
A tick bite at 22 resolves cleanly with doxycycline. Your body has abundant repair resources. The immune system recalibrates. The tissue damage heals. Done.
The same bite at 62? The antibiotics still kill the bacteria. But your depleted stem cell reserve can't contain the immune dysfunction or repair the tissue damage as effectively.
The infection clears. The wreckage accumulates. The symptoms persist.
Stem cell therapy, in this frame, isn't exotic. It's replenishment.
What Treatment at Our Clinic Looks Like
The procedure is notably undramatic. About 60–90 minutes. Most patients tell us they expected something far more intense.
No bone marrow aspiration. No fat tissue harvesting. We use pre-screened donor MSCs combined with platelet-rich plasma (PRP), delivered through a standard IV infusion. Patients describe a brief pinch from the needle, then nothing.
Here's exactly what happens:
- Pre-treatment review: Our medical team goes through your Lyme disease history, current symptom profile, and immune function assessment
- IV placement: Standard intravenous line, same as any IV medication
- Infusion: 50 million MSCs combined with PRP, delivered over 1–2 hours
- Post-infusion monitoring: Brief observation period, then you go home
What recovery looks like:
- 48–72 hours: Some fatigue is normal. That's the immune modulation kicking in.
- Week 1: Avoid anti-inflammatory meds (they interfere with what the MSCs are doing). Most patients return to normal activities in 2–3 days.
- Weeks 2–4: We begin supportive immune therapies. Some patients notice early improvements in energy or cognitive clarity during this window.
- Months 3–6: This is where the most significant changes tend to emerge. Peak benefits typically appear here.
We don't just hand you stem cells and send you home. Every patient receives a personalized immune support protocol, nutritional guidance, and ongoing monitoring. We track your progress because—frankly—we want the data. The more we learn about how our patients respond, the better we can refine this for everyone who comes after.
Investment
Complete Treatment Package: USD $8,000 – $12,000
That covers:
- Comprehensive Lyme disease evaluation and immune function assessment
- 50 million mesenchymal stem cells plus PRP
- Blood work and immune marker analysis
- The infusion procedure and all medical supplies
- Personalized immune support program
- Nutritional supplementation guidance
- 24/7 support during recovery
We'll be honest—this isn't cheap. But when you compare it to years of cycling through medications that manage symptoms without addressing the underlying dysfunction, the calculus changes for a lot of patients.
Safety and Honest Limitations
The safety profile is favorable. The 2024 Journal of Translational Medicine meta-analysis confirmed no increased adverse events across multiple controlled trials.
Common effects: mild infusion-site inflammation, occasional low-grade fever or headache in the first 24–48 hours, temporary fatigue.
One important caution. A 2023 case report in Emerging Infectious Diseases documented serious infection from improperly handled stem cell products during medical tourism. This is why facility quality matters enormously. All donor cells we use undergo rigorous testing for infections and genetic alterations, with sterility maintained throughout handling and administration. If you're evaluating clinics, ask hard questions about their cell sourcing and handling protocols. Any clinic that gets defensive about those questions is a red flag.
And here's the part we tell every patient in their consultation:
- Stem cell therapy does not cure Lyme disease.
- It does not replace antibiotics for active infection.
- Lyme-specific clinical trials are still in early stages.
- Individual results vary. Some patients improve significantly. Some don't.
We'd rather you walk in with realistic expectations than walk out disappointed. The goal isn't a miracle. The goal is giving your immune system the tools to stop fighting itself—and giving your body a real shot at repair.
Who This Is For
Based on the patients we've treated, stem cell therapy tends to be most appropriate for people who:
- Have completed antibiotic treatment but still experience persistent symptoms—fatigue, joint pain, brain fog, neurological issues
- Have been dealing with chronic symptoms for months or years without meaningful improvement
- Are looking for an approach that targets the underlying immune dysfunction rather than managing symptoms one by one
- Are in overall health that supports the procedure (we assess this during consultation)
If that sounds like you, it's worth a conversation.
Final Thoughts
Here's where we land.
The biology of PTLDS is becoming clearer by the year. We now know it's driven by immune dysregulation, persistent bacterial remnants, and chronic inflammation—not by active infection. That reframing matters, because it opens the door to interventions that target the actual problem rather than a bacterium that's already gone.
MSC therapy is one of those interventions. The mechanistic rationale is strong. The evidence from adjacent inflammatory and autoimmune conditions is substantial. The direct evidence for Lyme is early but growing.
Is it a sure thing? No. Nothing in medicine is.
But for the millions of people stuck in the gap between "your infection is cured" and "I still feel terrible"—a treatment that addresses the underlying immune dysfunction, rather than just managing symptoms, is worth understanding. And for many of our patients, it's been worth trying.
We built this program because we got tired of watching PTLDS patients run out of options. If you're in that place right now, we'd welcome the chance to talk through whether this approach makes sense for your situation.
Individual results may vary. No treatment outcomes are guaranteed. This information is for educational purposes only and should not replace professional medical advice. Statements not evaluated by the FDA or COFEPRIS.
Treatment
- Medical consultation
- 300 million stem cell IV + 10 billion exosomes
- 50 million stem cells via intrathecal injection
- Super immune IV + Detox IV + other as needed.
- Full blood panel
- Recommended supplement to take.
- Recommendation on things to do to enhance wellness.
